
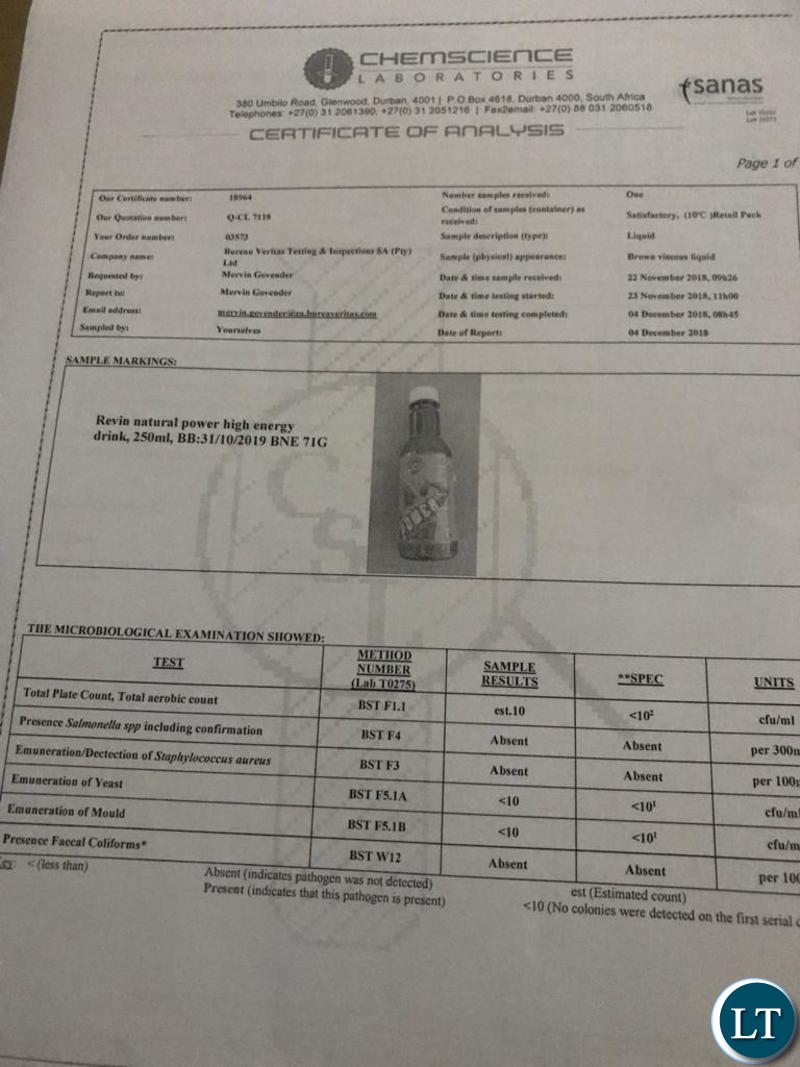
Various laboratory tests carried out on Natural Power SX Energy Drink have confirmed that product is safe for human consumption.
This is according to test results obtained from the Zambia Bureau of Standards, the Uganda National Bureau of Standards, the Malawi Bureau of Standards and the South African National Accreditation System (SANAS), a public entity which is recognized as the single National Accreditation Body in that country.
The Power High Energy produced by Revin Zambia Limited, a Ndola based company has come under spotlight after a Ugandan national allegedly suffered a six-hour erection after consuming the soft drink.
The complainant reportedly also suffered an abnormal heart beat with constant sweating prompting the Uganda National Drug Authority to issue a red flag on Natural Power SX Energy Drink.
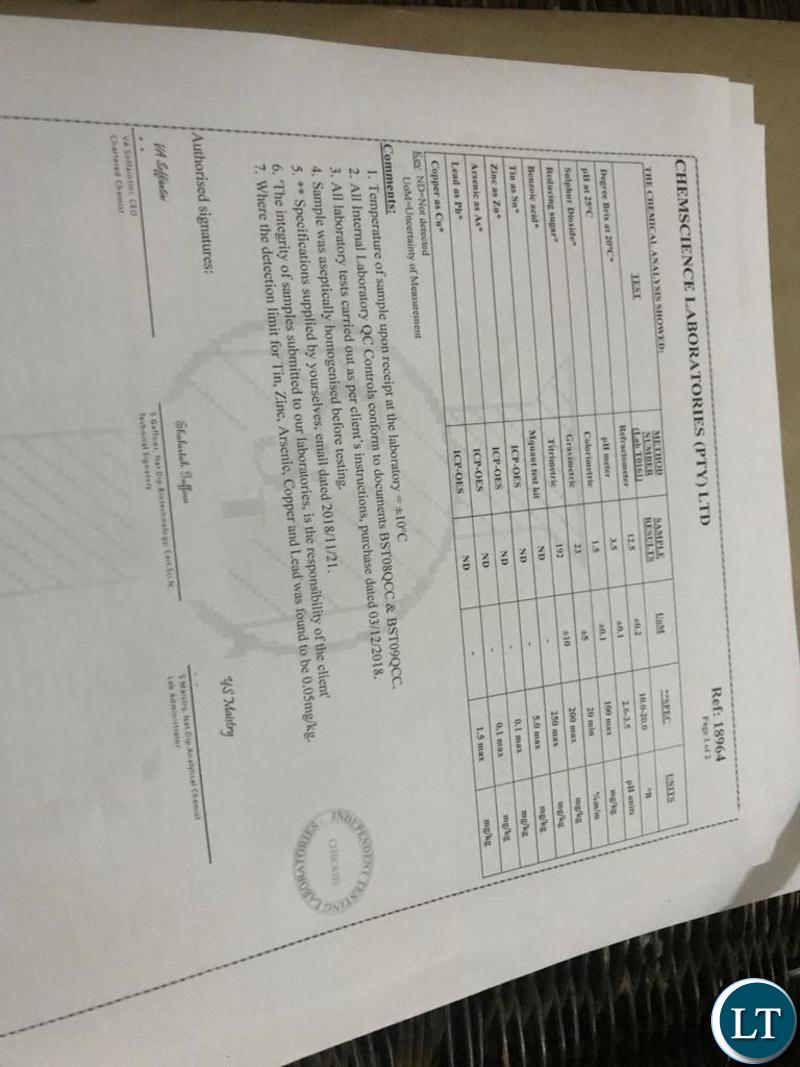
But an analysis of test results from various accreditation authorities in the SADC region have confirmed that contrary to the recent reports, Natural Power SX Energy Drink is safe and the product confirms to quality requirements.
In Malawi, test results carried out on 1st December 2015 showed that Natural Power SX Energy Drink complied with the MS 747 specifications and was subsequently allowed to be imported into that country.
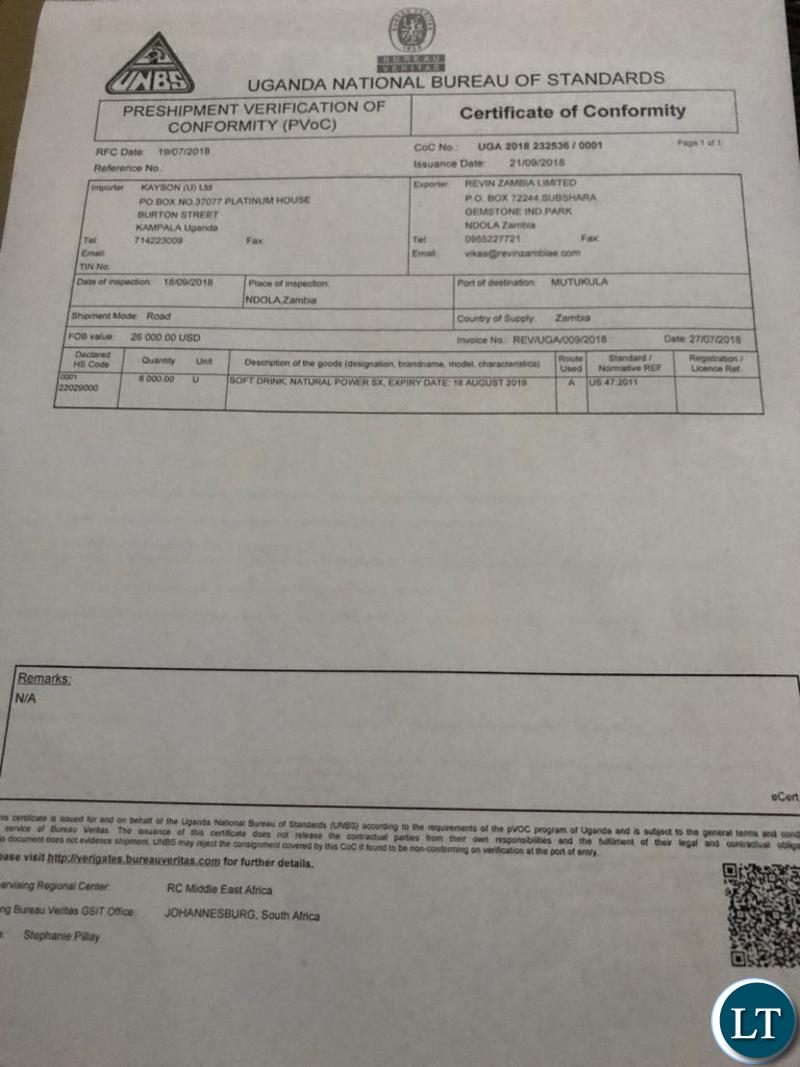
In Uganda, the product was issued with a Certificate of Conformity by the Uganda National Bureau of Standards on 19th August 2018 after it passed all the laboratory tests.
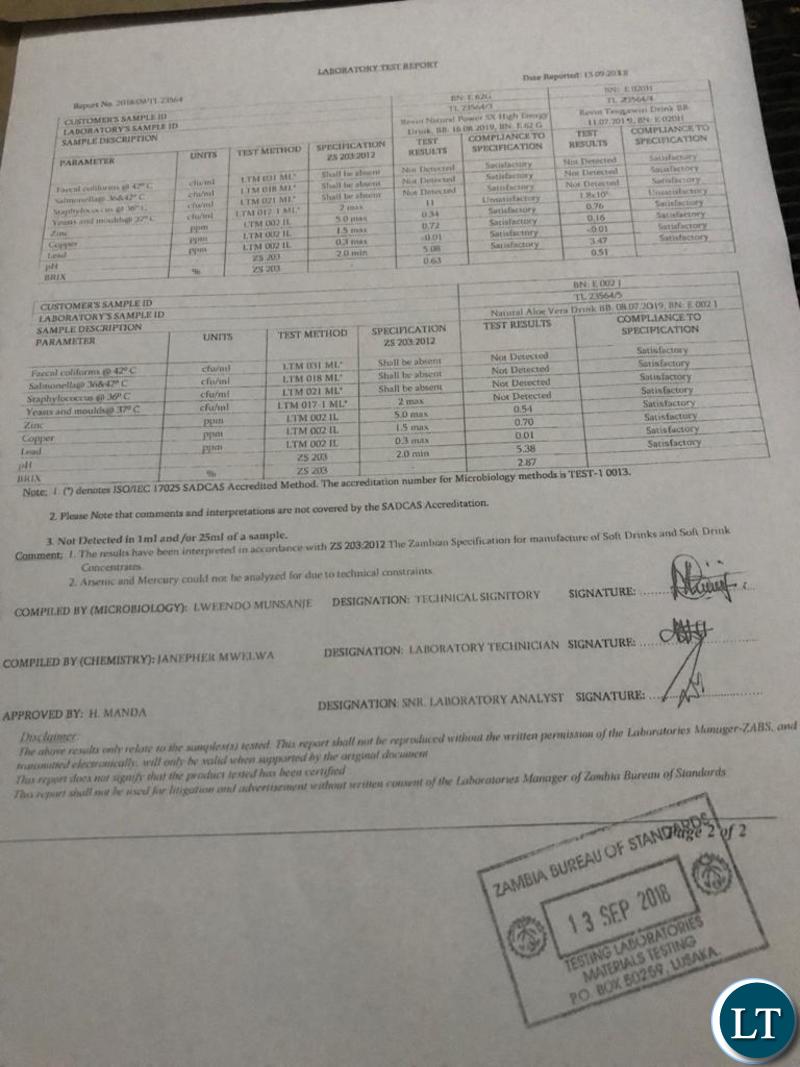
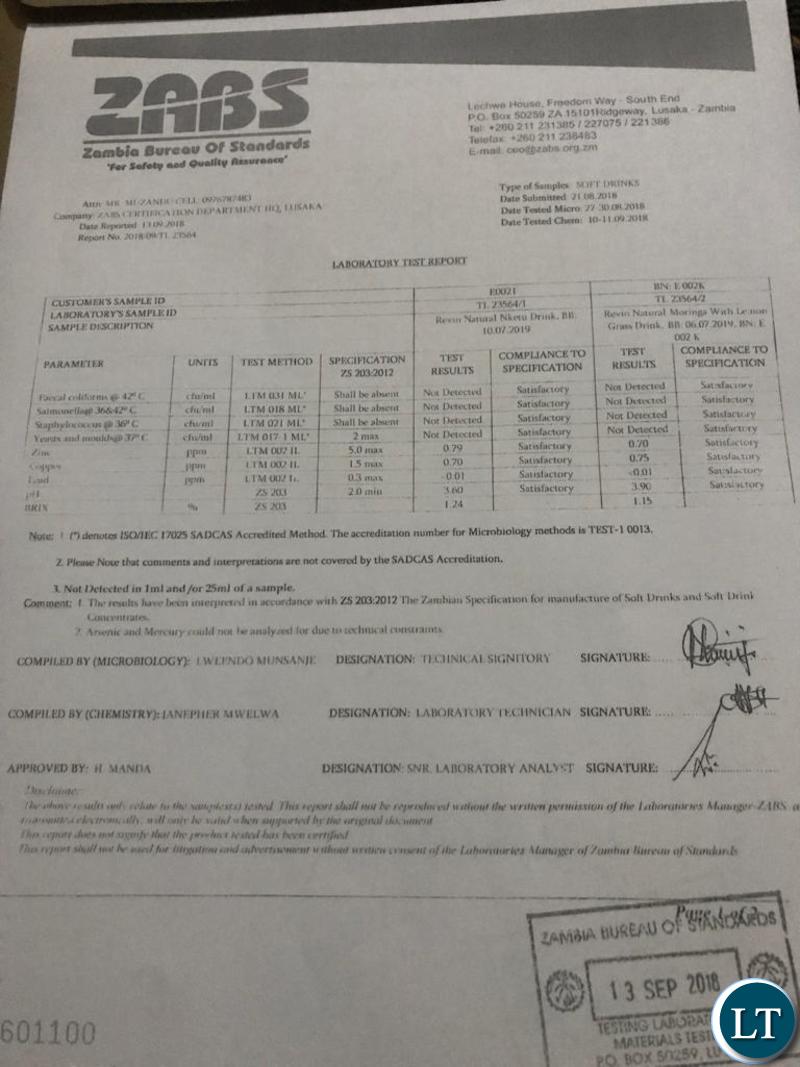
As for Zambia, Natural Power SX Energy Drink was found to be in compliance with ZS 2032012, the Zambian specifications for manufacture of soft drinks and soft drinks concentrates issued by the Zambia Bureau of Standards.
The company has also received huge export orders from Zimbabwe and Mozambique which some company insiders feel could be under threat if the allegations against the company persists.
And information has emerged that the incident in Uganda where a complaint was filed against Natural Power SX Energy Drink could be part of a scheme by business rivals to discredit the company and halt its regional expansion programme.
Some company insiders at Revin Zambia Limited who opted to remain anonymous have revealed in separate interviews that some Zambian business persons who are in similar lines of business have felt threatened by the growth of the company and have been scheming to bring it down.
“This Uganda incident is part of a grand plan to shut us down. We know our enemies have been planning for over three months to close us down and discrediting our most popular product is one of their plans,” the source said.
“What is interesting to note is that this particular product has been around in Zambia for over a year and there has never been any single complaint received, the same could be said about Malawi and even in the Congo DR where the product is very popular. This product is purely herbal and has no harmful side effects,” the company source said.
Further investigations revealed that Revin Zambia Limited has invested around 3.5 million dollars in its operations and employs over 54 full time workers with a major expansion programme lined up between 2020 and 2021.
Another 3.5 million dollars is earmarked to be invested in expanding the plant which will create a further 100 direct jobs with 300 indirect jobs by 2021.
Revin Zambia Limited also produces other similar products such as Fruit Juices and Tangawizi.






What did the Ugandans test recently? The drink (likely contaminated) that caused the erection or an unopened drink from a supermarket? Reporting on such matters must be done in deliberately detailed fashion so that we rule out the fact that most people who drink energy drinks also add their own concoctions before going on a romp.
Well these products are produced in batches and there could be batches that are spiked with viagra. These manufactures are manipulative and more so that our public institutions are now manned by PF political cadres and not professionals. The Ugandans will prove that the sample they tested has viagra in it.
Anything is possible with drinks made in Zambia especially under PF, people are desperate of money
The mediocrity of doing business in Africa is astounding. Whose idea was it from the company to publish test results from 2015? And from Malawi? Malawi is the equivalent of Dundumwezi. Also I don’t know why this company is panicking. Watch this product run off the shelves because now African men are aware that there is Viagra in it. Some will even increase the number of mistresses because of this drink. I wish I could buy shares in this company right now.
Ati soft drink… Should be hard drink kikikiki
This company needs a new Marketing Manager with a completely new team….to capitalize on all these Certificates of Conformity. I wonder where our utterly useless ministry of Religion stands on this issue?
Zambia the ‘Christian nation’ excels at exporting beer and sex libido at it neighbours…really laughable!!
@Ndobo you are wrong bro, apologize.
1. That booster is just like any libido booster, don’t take before get assured of availability of fit-woman. Not the mentally retarded. You never know the reaction.
2. That Zambian product is energy booster, need use when have something to do, not before going to bed, just like redbull.
3. Chibuku Shake-shake add some sugar, used to give me unbelievable libido, 4-6 hours non-stop. Believe it or not. In other words, us humans are different.
Ugandan politician was just a lucky guy.
How about the VIAGRA chemicals in your drinks?
Breach of property rights and mis-informing the public.
BAN IT!!!
Use the marijuana herb instead.
Sometimes I would Trump goes in 2020. But when you look at Dem comedians and level of their debate you just say Hey Let Trump continue.
They are childish inconsistent and directionless.
Same applies to Zambia.
Am a non partisan political consultant
clearly u are not
just because it’s from Zambia, there are lot of harmful products from South Africa why not ban there use inZambia. and you are the same people wanting to legalize GMOs which have profound effect to the human body than this Power energy.
I am waiting for the day when skin bleaching will be banned and silly wigs and weaves by civil servants especially ZP and Army.
Comment:the man must have used other concoction,or he is allergic to it.Not every meds works well to every individual so the man’s case is not different
I thought thats what he wanted by taking the drink! Stop complaining and enjoy its use after all no one forced him to take it.
I believe that it is imperative that within such a large company that is expanding across such African countries, that there could always be room for error. So far there has only been one case as of such, and the company complied with the governments and showed the results of all their products. Forcing a ban on such a hot product would be as if you shut down a bank for mis-printing one currency note.
This situation should be tackled by acting smart, not acting quick. Let us wait for more information to be revealed about the case before taking a stance on this case.
While I get your point, what an inappropriate example you have used of ‘a bank misprinting one currency note’. Banks do NOT print currency!
This kind of ignorance is astounding!!! Indeed knowledge is power. When professionals speak, please listen, but alas, here it appears the empty tins speak the loudest. These simple basic microbiological tests you have shown us here are important but am afraid very, very, very, very simple analytical methods used to detect whether or not there’s presence of (a targeted list) fungus, viruses, bacteria etc that might cause food poisoning and diseases. So simple that they cannot detect adulteration of drugs (APIs). You need to do a full monograph analysis using known or reference standards such as USP, BP etc. This should give you an idea whether or not ZABs has both the equipment and technical competence to carry out this and detect Viagra or any API in a drink. These results are so basic…
Only one case out of millions sold looks
A bit tricky.just for benefit of doubt wait
For further complaints.
Ba ku chipata Bakamba ati Nvubwe meaning approdiatcs.
Probably the Ugandan took overdose and to make matters worse the expected woman failed to turn up.
I think what happened is that the man may have been underperforming, from the girlfriend’s perspective. So, she put up a plan to spike his drink with Sildenafil Citrate (Viagra) – and the results were obvious. This case needs further investigation – who was he with? Was this the first time he took this drink? The nonsense from some bloggers here “PF this PF that” is absolute id.iocy.
The president for psz clearly stated theres viagra in the drink. You need to test for that. Instead of wanting to do damage control so that the company can continue working. Zambia che. How much were u paid?
am still wondering,why 1 person in a million consumers only experienced such reactions in his body,
I’ve consumed this drink several times, but av never experienced such reactions.
The complainant must give full proof,maybe he’s allergic????????????